ASO-301
ASCORBATE OXIDASE from Cucumis sp.
PREPARATION and SPECIFICATION
| Appearance | Light blue amorphous powder, lyophilized | |
|---|---|---|
| Activity | GradeⅢ 200U/mg-solid or more (containing approx. 70% of stabilizers) |
|
| Contaminants | Catalase | ≤1.0×10-1% |
| Phosphatase | ≤2.0×10-2% | |
| Stabilizers | BSA, borax, basic amino acids. | |
PROPERTIES
| Stability | Stable at −20℃ for at least one year(Fig.1) |
|---|---|
| Molecular weight | 132,000 1), 140,000 2) |
| Isoelectric point | between 6.0 and 7.8 1), 8.2 2) |
| Michaelis constant | 2.5×10-4M (Ascorbate) |
| Structure | 8 copper atoms per enzyme molecule 1,2) |
| Inhibitors | cyanide, Na2S, diethyldithiocarbamate (Na) |
| Optimum pH | 5.6(Fig.4) |
| Optimum temperature | approx. 30℃(Fig.5) |
| pH Stability | pH7.0−10.0 (25℃, 17hr)(Fig.6) |
| Thermal stability | below 40℃ (pH 8.0, 30min)(Fig.7) |
| Substrate specificity | This enzyme oxidizes ascorbic acid and several ascorbic derivatives.3) |
| Effect of various chemicals | (Table 1) |
APPLICATIONS
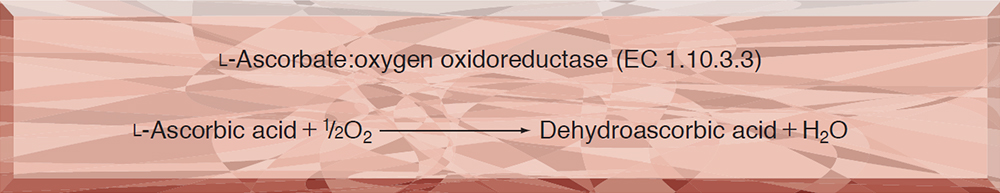
This enzyme is useful for enzymatic determination of ascorbic acid and for eliminating the interference of ascorbic acid in clinical analysis.
ASSAY
Principle
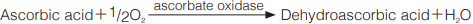
The disappearance of ascorbic acid is measured at 245nm by spectrophotometry.
Unit definition
One unit causes the decrease of one micromole of ascorbic acid per minute under the conditions described below.
Method
Reagents
| A. Ascorbic acid solution | 1.0mM [Dilute the stock solution (10mM) to 10-fold volume with 0.2 M KH2PO4 solution containing 1.0mM EDTA.](Prepare freshly) Stock solution : 176mg L-ascorbic acid (MW=176.13)/100ml of 1.0mM HCl solution containing 1.0mM EDTA (Stable for one month if stored at 0−5℃) |
|---|---|
| B. Na2HPO4 solution | 10mM |
| C. HCl solution | 0.2N |
| D. Enzyme diluent | 10mM Na2HPO4 solution containing 0.05% BSA (Prepare freshly) |
Procedure
1.Prepare the following reaction mixture in a test tube and equilibrate at 30℃ for about 5 minutes.
| 0.5ml | Substrate solution | (A) |
|---|---|---|
| 0.5ml | Na2HPO4 solution | (B) |
| (pH of the reaction mixture should be 5.6.) | ||
| Concentration in assay mixture | |
|---|---|
| KH2PO4 | 82 mM |
| Na2HPO4 | 5.5 mM |
| Ascorbic acid | 0.45 mM |
| EDTA | 0.45 mM |
| BSA | 45.4μg/ml |
2.Add 0.1ml of the enzyme solution* and mix.
3.After exactly 5 minutes at 30℃, add 3.0ml of HCl solution (C) to stop the reaction and measure the optical density at 245nm against water (OD test).
At the same time, prepare the blank by first mixing the reaction mixture with 3.0ml of HCl solution (C) after 5 min-incubation at 30℃, followed by addition of the enzyme solution (OD blank).
*Dissolve the enzyme preparation in ice-cold distilled water (more than 60U/ml) and dilute to 0.15−0.25U/ml with ice-cold enzyme diluent (D), immediately before assay.
Calculation
Activity can be calculated by using the following formula :
Volume activity (U/ml) =
-
ΔOD (OD blank−OD test)×Vt×df
10.0×1.0×t×Vs
= ΔOD×0.820×df
Weight activity (U/mg) = (U/ml)×1/C
| Vt | : Total volume (4.1ml) |
| Vs | : Sample volume (0.1ml) |
| 10.0 | : Millimolar extinction coefficient of ascorbic acid under the assay condition at pH 1.0 (cm2/micromole) |
| 1.0 | : Light path length (cm) |
| t | : Reaction time (5 minutes) |
| df | : Dilution factor |
| C | : Enzyme concentration in dissolution (c mg/ml) |
REFERENCES
1)T.Nakamura, N.Makino and Y.Ogura; J.Biochem., 64,189 (1968).
2)V.Ts.Aikazyan and R.M.Nalbandyan; FEBS LETTERS, 104,127 (1979).
3)G.A.White and F.G.Smith; Nature, 190,187 (1961).
Table 1. Effect of Various Chemicals on Ascorbate oxidase
[The enzyme dissolved in distilled water (60U/ml) was incubated with each chemical at 25℃ for 1hr.]
-
Chemical Concn.(mM) Residual activity(%) None - 100 Metal salt 2.0 MgCl2 88 CaCl2 86 Ba(OAc)2 86 FeCl3 34 CoCl2 83 MnCl2 88 ZnCl2 90 CdCl2 87 NiCl2 79 CuSO4 90 Pb(OAc)2 91 AgNO3 3.7 HgCl2 42 2-Mercaptoethanol 2.0 75 PCMB 1.0 26 -
Chemical Concn.(mM) Residual activity(%) MIA 2.0 3.5 NEM 2.0 75 IAA 2.0 21 Hydroxylamine 2.0 81 EDTA 5.0 80 o-Phenanthroline 2.0 50 α,α′-Dipyridyl 1.0 78 Borate 50 75 NaF 2.0 84 NaN3 2.0 85 Triton X-100 0.10% 84 Brij 35 0.10% 24 Tween 20 0.10% 19 Span 20 0.10% 97 Na-cholate 0.10% 80 SDS 0.05% 83 DAC 0.05% 88
EDTA, Ethylenediaminetetraacetate; SDS, Sodium dodecyl sulfate; DAC, Dimethylbenzylalkylammonium chloride.
-
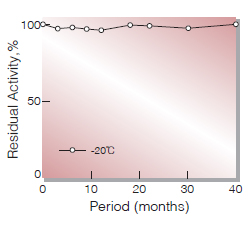
Fig.1. Stability (Powder form)
(kept under dry conditions)
-
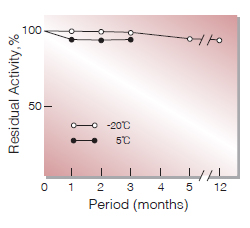
Fig.2. Stability (Powder form)
(kept under dry conditions)
-
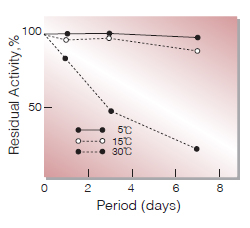
Fig.3. Stability (Liquid form)
enzyme concn. :8,000U/ml buffer composition:45mM borate buffer,pH7.8
-
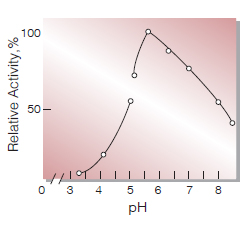
Fig.4. pH-Activity
30℃ in 0.33 M buffer solution: pH3.0-6.0,acetate; pH5.0-8.0, phosphate
-
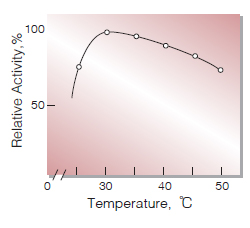
Fig.5. Temperature activity
(in 0.33M phosphate buffer,pH5.6)
-
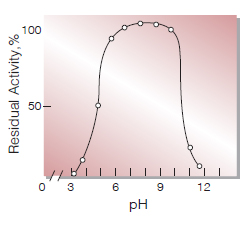
Fig.6. pH-Stability
25℃, 17hr-treatment with Britton-Robinson's buffer
-
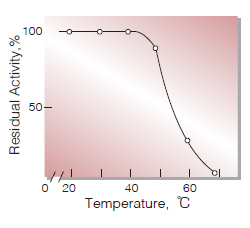
Fig.7. Thermal stability
30min-treatment with 50mM phosphate buffer,pH 8.0 enzyme concn.: 12U/ml
活性測定法(Japanese)
1. 原理
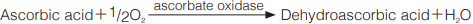
Ascorbic acidの消失量を245nmの吸光度の変化で測定する。
2.定義
下記条件下で1分間に1マイクロモルのアスコルビン酸を酸化する酵素量を1単位(U)とする。
3.試薬
- 1.0mMアスコルビン酸溶液〔保存溶液(10mMのL-アスコルビン酸溶液)を1.0mM EDTAを含む0.2M KH2PO4溶液で10倍希釈する〕(用時調製)
保存溶液は176mgのL-アスコルビン酸(試薬特級,MW=176.13)を精秤し1.0mM EDTAを含む1.0mM HCI溶液100mlに溶解して調製する(0~5℃保存で1ケ月は使用可能)。 - 10mM Na2HPO4溶液
- 0.2N HCl溶液
酵素溶液:酵素標品を予め氷冷した蒸留水で溶解(60U/ml以上)し、分析直前に0.05%BSAを含む10mM Na2HPO4溶液(氷冷)で0.15~0.25U/mlに希釈する。
4.手順
1.試験管に下記反応混液を調製し,30℃で約5分間予備加温する。
| 0.5ml | 基質溶液 | (A) |
| 0.5ml | Na2HPO4溶液 | (B) |
| (反応混液のpHは5.6) | ||
2.酵素溶液0.1mlを加え,反応を開始する。
3.30℃で正確に5分間反応させた後,HCl溶液(C)3.0mlを加えて反応を停止させる。この液につき245nmにおける吸光度を測定する(ODtest)。
4.盲検は反応混液1を30℃で5分間放置後, HCl溶液(C)3.0mlを加えて混和し,次いで酵素溶液0.1mlを加えて調製する。以下同様に吸光度を測定する(ODblank)。
5.計算式
U/ml =
-
ΔOD(OD blank−OD test)×4.1(ml)×希釈倍率
10.0×1.0×5(分)×0.1(ml)
| = ΔOD×0.82×希釈倍率 | |
| U/mg | = U/ml×1/C |
| 10.0 | : アスコルビン酸の上記測定条件下(pH1.0)でのミリモル分子吸光係数 (cm2/micromole) |
| 1.0 | : 光路長(cm) |
| C | : 溶解時の酵素濃度(c mg/ml) |
CONTACT
お問い合わせ-
各種製品に関するご質問・ご相談はこちらよりお問い合わせください。
