-
GLO-101・201
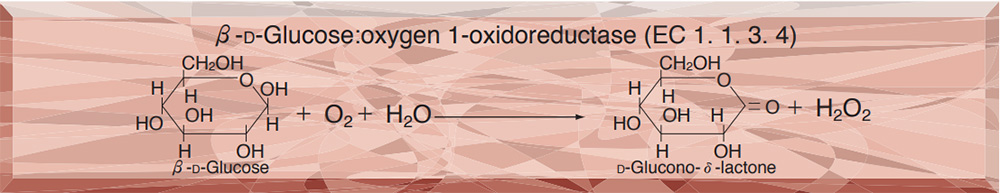
GLUCOSE OXIDASE from Aspergillus sp.
PREPARATION and SPECIFICATION
| Appearance | Yellowish amorphous powder, lyophilized | ||
|---|---|---|---|
| Activity | GradeⅠ | 180U/mg-solid or more | |
| GradeⅡ | 100U/mg-solid or more | ||
| (containing approx. 50% of stabilizers) | |||
| Contaminant | Catalase | GradeⅠ | ≤5.0×10-3% |
| GradeⅡ | ≤3.0% | ||
| Stabilizers | Potassium gluconate, sodium glutamate | ||
PROPERTIES
| Stability | Stable at −20℃ for at least one year(Fig.1) | |
|---|---|---|
| Molecular weight | approx. 153,000 | |
| Michaelis constants | 3.3×10-2M (β-D-Glucose),3) | |
| 6.1×10-2M (2-Deoxyglucose) | ||
| Structure | Glycoprotein with 2 moles of FAD | |
| Inhibitors | p-Chloromercuribenzoate, heavy metal ions (Cu++, Hg++, Ag+) | |
| Optimum pH | 4.5(Fig.3) | |
| Optimum temperature | 40−50℃(Fig.4) | |
| pH Stability | pH 4.5−6.0 (30℃, 20hr)(Fig.5) | |
| Thermal stability | below 50℃ (pH 5.7, 1hr)(Fig.6) | |
| Substrate specificty | (Table 1) | |
| Effect of various chemicals | (Table 2) | |
APPLICATIONS
This enzyme is useful for enzymatic determination of glucose, and for amylase-activity assay when coupled with α-glucosidase (AGH-211, if maltooligosaccharide or modified starch is used as a substrate) in clinical analysis.
ASSAY
Principle
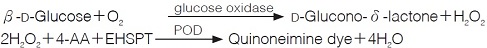
4-AA : 4-Aminoantipyrine
EHSPT : N-Ethyl-N-(2-hydroxy-3-sulfopropyl)-m-toluidine
The appearance of quinoneimine dye is measured at 555nm by spectrophotometry.
Unit definition
One unit causes the formation of one micromole of hydrogen peroxide (half a micromole of quinoneimine dye) per minute under the conditions described below.
Method
Reagents
| A. MES-Na buffer pH 5.7 | 0.1M[Dissolve 2.13g of 2-(N-morpholino) ethansulfonic acid (MW=213.25) in ca. 60ml of H2O and, after adjusting the pH to 5.7 with 1N NaOH at 25℃, fill up to 100ml with H2O](Stable at 5℃ for one month) | |
|---|---|---|
| B. Glucose solution | 15%[Dissolve 1.5g of β-D-glucose and fill up to 10ml with H2O]for at least 2hrs before assay.) (Should prepare fresh) | |
| C. 4-AA solution | 0.5%[50mg fo 4-aminoantipyrine (MW=203.25)/10ml of H2O](Stable at 5℃ in a brownish bottle for at least one week) | |
| D. EHSPT solution | 40mM[118mg of N-Ethyl-N-(2-hydroxy-3-sulfopropyl)-m-toluidine (MW=295.3)/10ml of H2O] (Stable at 5℃ in a brownish bottle for at least one week) | |
| E. Peroxidase solution | 500U (purpurogalin unit)/ml of H2O | |
| F. Enzyme diluent | 10mM MES-Na buffer, pH 5.7, containing 0.1% Triton X-100 | |
Procedure
1. Prepare the following working solution in a brownish bottle and store on ice.
| (Should be prepared fresh) | ||
| 30 ml | Buffer solution | (A) |
| 6 ml | Substrate solution | (B) |
| 0.3ml | 4-AA solution | (C) |
| 0.3ml | EHSPT solution | (D) |
| 0.3ml | POD solution | (E) |
| Concentration in assay mixture | |
|---|---|
| MES buffer | 79 mM |
| D-Glucose | 131 mM |
| 4-AA | 0.2mM |
| EHSPT | 0.3mM |
| POD | ca.4 U/ml |
2. Pipette 3.0ml of working solution into a cuvette (d=1.0cm) and equilibrate at 37℃ for about 5 minutes.
3. Add 0.1ml of the enzyme solution* and mix by gentle inversion.
4. Record the increase in optical density at 555nm against water for 2 to 3 minutes in a spectrophotometer thermostated at 37℃, and calculate the ΔOD per minute from the initial linear portion of the curve (ΔOD test).
At the same time, measure the blank rate (ΔOD blank) by using the same method as the test except that enzyme diluent (F) is added instead of the enzyme solution.
*Dissolve the enzyme preparation in ice cold enzyme diluent (F) and dilute to 0.05−0.2U/ml with the same buffer, immediately before the assay.
Calculation
Activity can be calculated by using the following formula :
-
Volume activity (U/ml) =
-
ΔOD/min (ΔOD test−ΔOD blank)×Vt×df
32.8×1/2×1.0×Vs
-
= ΔOD/min×1.89×df
Weight activity (U/mg) = (U/ml)×1/C
| Vt | : Total volume (3.1ml) |
| Vs | : Sample volume (0.1ml) |
| 32.8 | : Millimolar extinction coefficient of quinoneimine dye under the assay conditions (cm2/micromole) |
| 1/2 | : Factor based on the fact that one mole of H2O2 produces a half of quinoneimine dye. |
| 1.0 | : Light path length (cm) |
| df | : Dilution factor |
| C | : Enzyme concentration in dissolution (c mg/ml) |
REFERENCES
1) The Enzymes, Vol.ⅫB, P.421 (P.D.Boyer, ed.), Academic Press (1975).
2) Method in Enzymology, Vol.Ⅸ, p.82 (S.P.Colowick and N.O.Kaplan, ed.),Academic Press (1966).
3) B.E.P.Swoboda and V.Massay; J.Biol.Chem., 240, 2209 (1965).
4) P.J.Auses, S.L.Cook and J.T.Maloy; Anal.Chem., 47, 244 (1975).
5) D.C.Williams, G.F.Huff and W.R.Gaitz; Clin.Chem., 22, 372 (1976).
Table 1. Substrate Specificity of Glucose oxidase
[0.1M of Substrate, 79mM MES buffer, pH 5.7, at 30℃ ]
-
Substrate (0.1M) Relative activity(%) D-Glucose 100 2-Dexy-D-glucose 16.2 Glucono-1,5-lactone 0.06 L-Glucose 0.00 Galactose 3.10 Mannose 2.10 -
Substrate (0.1M) Relative activity(%) Fructose 0.24 Xylose 0.93 Ribose 0.00 Maltose 0.69 Lactose 0.00
Table 2. Effect of Various Chemicals on Glucose oxidase
[The enzyme dissolved in 0.1M MES buffer, pH 5.7 (10U/ml) was incubated with each chemical for 2hr at 25℃.]
-
Chemical Concn.(mM) Residual
activity(%)None - 100 Metal salt 2.0 MgCl2 92.6 CaCl2 93.6 BaCl2 94.4 CoCl2 98.1 MnCl2 95.1 ZnSO2 94.3 FeCl2 96.8 NiCl2 91.7 CuSO4 71.6 AgNO3 58.6 HgCl2 0.7 PCMB 2.0 31.6 MIA 2.0 96.8 NaF 2.0 97.1 -
Chemical Concn.(mM) Residual
activity(%)NaN3 20 96.3 EDTA 5.0 97.3 o-Phenanthroline 2.0 95.3 α,α′-Dipyridyl 2.0 99.5 Borate 50.0 96.1 IAA 2.0 96.1 MIA 2.0 101.1 Hydroxylamine 10.0 98.3 Sodium bisulfite 10.0 100.0 hydrazine 10.0 103.1 Triton X-100 0.1% 111.2 Brij 35 0.1% 108.0 Tween 20 0.1% 110.7 Span 20 0.1% 106.7 Na-Cholate 0.1% 106.1 SDS 0.1% 113.1
PCMB, p-Chloromercuribenzoate; MIA, Monoiodoacetate; EDTA, Ethylenediamimetetraacetate; IAA, Iodoacetamide; NEM, N-Ethylmaleimide; SDS, Sodium dodecyl sulfate.
-
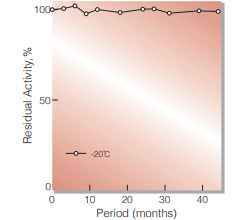
Fig.1. Stability (Powder form)
(kept under dry conditions)
-
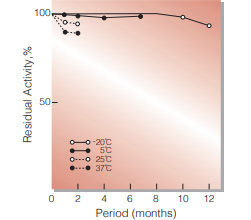
Fig.2. Stability (Powder form)
(kept under dry conditions)
-
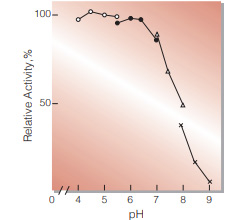
Fig.3. pH-Activity
37℃,5min-reaction in 79mM buffer solution : ○̶̶○ . acetate;●̶̶● MES;△̶̶△, BES;×̶̶×, BICINE
-
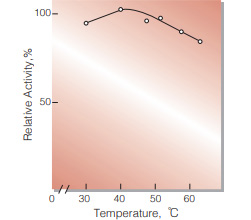
Fig.4. Temperature activity
(5min-reaction in 79mM MES buffer, pH5.7)
-
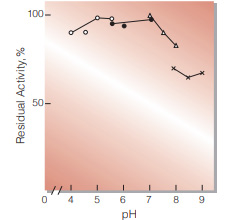
Fig.5. pH-Stability
30℃,20hr-treatment with 0.1M buffer solution : ○̶̶○ . acetate : ●̶̶● MES : △̶̶△, BES : ×̶̶×, BICINE
-
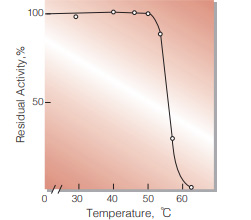
Fig.6. Thermal stability
(1hr-treatment in 79mM MES buffer , pH5.7)
活性測定法(Japanese)
1. 原理
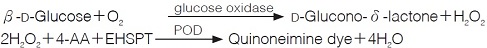
4-AminoantipyrineとEHSPTの酸化縮合生成物であるQuinoneimine色素を555nmで測定し,上記反応で生成したH2O2量を定量する。
2.定義
下記条件下,1分間に1マイクロモルのH2O2を生成する酵素量を1単位 (U)とする。
3.試薬
- 0.1M MES-Na緩衝液,pH5.7 (5℃保存で1カ月間使用可能)
- 15%グルコース溶液 (使用前に2時間以上放置する)(用時調製)
- 0.5% 4-AA溶液 (褐色瓶中5℃保存で1週間安定使用可能)
- 40mM EHSPT (TOOS)溶液 (褐色瓶中5℃保存で1週間安定使用可能)
- 500PU/ml POD溶液
酵素溶液:酵素標品を予め氷冷した0.1% Triton X100を含む10mM MES-Na緩衝液 (A)で溶解し,分析直前に同緩衝液で0.05〜0.2U/mlに希釈する。
4.手順
1.褐色瓶中に下記反応混液を調製し氷冷保存する。
| (用時調製) | ||
| 30 ml | MES-Na緩衝液 | (A) |
| 6 ml | 基質溶液 | (B) |
| 0.3ml | 4-AA水溶液 | (C) |
| 0.3ml | EHSPT(TOOS)水溶液 | (D) |
| 0.3ml | POD水溶液 | (E) |
| (褐色瓶にて氷冷保存) | ||
2.反応混液3.0mlをキュベット(d=1.0cm)に分注し37℃で約5分間予備加温する。
3.酵素溶液0.1mlを添加しゆるやかに混和後,水を対照に37℃に制御された分光光度計で555nmの吸光度変化を2〜3分間記録し,その直線部分から1分間あたりの吸光度変化を求める(ΔOD test)。
4.盲検は反応混液①に酵素液の代りに酵素希釈液〔MES-Na緩衝液(A)〕0.1mlを加え,上記同様に操作を行い,1分間当りの吸光度変化を求める(ΔODblank)。
5.計算式
-
U/ml =
-
ΔOD/min (ΔOD test−ΔOD blank)×3.1×df
32.8×1/2×1.0×0.1
| = ΔOD/min×1.89×df | |
| U/mg | = U/ml×1/C |
| 32.8 | : Quinoneimine色素の上記測定条件下でのミリモル分子吸光係数(cm2/micromole) |
| 1/2 | : 酸素反応で生成したH2O2の1分子のから形成するQuinoneimine色素は1/2分子である事による係数 |
| 1.0 | : 光路長(cm) |
| C | : 溶解時の酵素濃度(c mg/ml) |
CONTACT
お問い合わせ-
各種製品に関するご質問・ご相談はこちらよりお問い合わせください。
